Antiretroviral medications
are used to suppress the reproduction of the HIV virus and slow progression of
HIV-related disease. Highly Active Antiretroviral Therapy (HAART) is the
treatment protocol that is recommended for HIV infection. HAART combines 3 or
more antiretroviral medications in a daily regimen. Antiretroviral medications are grouped into six drug classes according to how they fight HIV. The six classes are non-nucleoside reverse transcriptase inhibitors (NNRTIs), nucleoside reverse transcriptase inhibitors (NRTIs), protease inhibitors (PIs), fusion inhibitors, CCR5 antagonists, and integrase inhibitors.
Recommended HIV treatment regimens include three or more antiretroviral medications from at least two different drug classes. Taking a combination of antiretroviral medications from different classes is the most effective way to control the virus. Some anti-HIV medications are available in combination (two or more medications in one pill).The exact medication regimen
is highly individualized, depending upon many factors. Usually taking only 1 or
2 medications is not effective. Unless the individual is taking 3 or more medications,
the decrease in viral load tends to be temporary.
Antiretroviral medications
There are 6 classes of
antiretroviral medication used in the treatment of HIV.
|
Class |
Generic
name |
Brand
name |
|
Nonnucleoside Reverse Transcriptase Inhibitors(NNRTIs) bind to
and disable reverse transcriptase, a protein that HIV needs to replicate.
|
Delavirdine | Rescriptor, DLV |
| Efavirenz | Sustiva, EFV | |
| Nevirapine |
Viramune, NVP | |
| Second Gen (NNRTI) | Etravirine | Intelence |
| Rilpivirine | Edurant | |
|
Nucleoside
Reverse Transcriptase Inhibitors (NRTIs) are faulty versions of building
blocks that HIV needs to replicate. When HIV uses an NRTI instead of a
normal building block, reproduction of the virus is stalled. |
Abacavir | Ziagen, ABC |
|
Abacavir Lamivudine |
Epzicom | |
| Abacavir, Lamivudine, Zidovudine |
Trizivir | |
| Didanosine | Videx, ddI, Videx EC | |
| Emtricitabine | Emtriva, FTC, Coviracil | |
| Emtricitabine, Tenofovir DF | Truvada | |
| Lamivudine | Epivir, 3TC | |
| Lamivudine, Zidovudine | Combivir | |
| Stavudine | Zerit, d4T | |
| Tenofovir DF | Viread, TDF | |
| Zalcitabine | Hivid, ddC | |
| Zidovudine | Retrovir, AZT, ZDV | |
|
Protease
Inhibitors (PIs) disable protease, a protein that HIV needs to replicate. |
Amprenavir | Agenerase, APV |
| Atazanavir | Reyataz, ATV |
|
| Fosamprenavir | Lexiva, FPV | |
| Indinavir | Crixivan, IDV | |
| Lopinavir, Ritonavir | Kaletra,LPV/r | |
| Nelfinavir | Viracept, NFV | |
|
Fusion
Inhibitors function by blocking HIV entry into cells. |
Enfuvirtide | Fuzeon, T-20 |
Chemokine receptor 5 (CCR5) antagonist selectively and reversibly binds to CCR5 inhibiting attachment and fusion |
Maraviroc | Selzentry |
| Integrase Inhibitors | Raltegravir | Isentress |
Instant Feedback:
Adjustments to Treatment
People who are receiving antiretroviral medications should have a baseline viral load and CD4 count done and regular evaluations during treatments. If the viral load increases and CD4 count decreases, the treatment regimen may need to be adjusted. In that case, the physician will evaluate 3 factors to determine what adjustments need to be made:
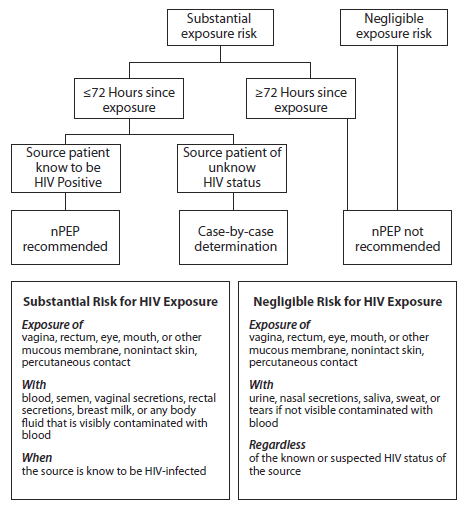
Non-occupational Post-exposure prophylaxis (nPEP)
Prophylaxis for HIV is a medical intervention designed to prevent HIV infection after exposure to the virus. Patients who have had sexual, injection-drug–use, or other nonoccupational exposures to potentially infectious fluids of persons known to be HIV infected are at risk for acquiring HIV infection and should be considered for nPEP if they seek treatment within 72 hours of exposure.
Overall, nPEP is more likely to be effective when the exposure is a single episode and nPEP is initiated within 72 hours of exposure. It is not appropriate for cases of multiple sexual exposures or injection drug use (IDU) exposures over time or for exposures that occurred >72 hours before starting nPEP treatment.
Antiretroviral Regimens for Nonoccupational Postexposure Prophylaxis of HIV Infection * |
|
| Protease Inhibitor Based |
|
| Integrase inhibitor based |
|
Occupational Post-exposure prophylaxis (nPEP)
In the work setting, HIV infection may occur through percutaneous injuries (e.g., needlesticks) or mucocutaneous exposures (e.g., mucous membrane or nonintact skin exposure to blood or other potentially infectious body fluids). The risk of HIV seroconversion after occupational exposure with an HIV-contaminated hollow-bore needle is best described as 0.3%, on average. Another way of describing this to an exposed HCW is that, without PEP, HIV transmission occurs about once in 300 instances of needlestick from a known HIV-infected source patient. In a retrospective case-control study of HCWs with percutaneous exposure to HIV, the following exposure and source patient factors were associated with an increased risk of HIV transmission:
Beginning Occupational PEP requires assessment of the: infection risk, adverse effects of antiretroviral therapy including potential fetal teratogenic effects and the patient commitment to completing a 28 day regimen.
Antiretroviral Regimens for Occupational Postexposure Prophylaxis of HIV Infection 2 NRTIs
- Tenofovir 300 mg once daily + emtricitabine 200 mg once daily (available as Truvada, 1 tablet once daily)
- Zidovudine 300 mg BID + lamivudine 150 mg BID (available as Combivir, 1 tablet BID)
Protease Inhibitors
- Lopinavir/ritonavir (Kaletra) 400/100 mg BID
- Atazanavir 300 mg once daily + ritonavir 100 mg once daily
- Darunavir 800 mg once daily + ritonavir 100 mg once daily
- Atazanavir 400 mg once daily
Integrase Inhibitors
- Raltegravir 400 mg BID
Follow-up for workers on 28 day PEP regimens
Instant Feedback:
nPEP is designed for treatment >72 hours after exposure to HIV.
Treatment during pregnancy
"Current U.S. Public Health Service guidelines recommend treating HIV infection in all pregnant women, using the same principles and modalities as used for nonpregnant individuals. HIV-infected pregnant women should receive potent combination antiretroviral therapy (ART) regimens comprising at least three antiretroviral medications (ARVs). The use of zidovudine (ZDV) prophylaxis alone (monotherapy) is not recommended but may be considered for the few women whose HIV RNA levels are <1,000 cells/µL on no ART and who decline combination ART. The choice of ARV regimen should be based on what is likely to be optimal for the woman's health, the potential effect on the fetus and infant, resistance test results, the woman's previous experience, if any, with ART, and her stage of pregnancy. ARV resistance testing should be performed before initiating or changing therapy. ZDV should be included in the regimens of all pregnant women unless there is severe toxicity or documented drug resistance. ARV agents with known teratogenic effects, such as efavirenz, should be avoided, especially in the first trimester. Women who do not need ART for their own health and are taking ART only for the prevention of perinatal transmission may delay initiation of therapy until after the first trimester. This is a period of rapid organogenesis, and there is an increased risk of birth defects if teratogen exposure occurs. For women already taking ART at the time they become pregnant, the regimen should be reevaluated for its appropriateness during pregnancy to avoid potentially toxic medications and to ensure maximal virologic suppression. The ART regimen may be changed if necessary, but should continue without interruption. Discontinuation of ART could lead to an increase in viral load, which could result in a decline in immune status and an acceleration of disease progression, thereby increasing the risk of HIV transmission to the fetus."*
Side effects of HIV treatment
Numerous antiretroviral drugs, have been approved by the FDA for treatment of HIV. Each of these drugs is associated with side effects. The primary method of treating the side effects is to make a change in the antiviral medications. Major side effects may include the following:
Hepatotoxicity: Women, especially those who are pregnant; those with existing liver diseases, such as Hepatitis B or C; and those who use alcohol may be at higher risk for developing hepatotoxicity.
Hyperglycemia: An increase in blood glucose levels by itself, or as part of diabetes mellitus, is a side effect of all protease inhibitors (PIs).
Hyperlipidemia: An increase in the lipids in the blood, such as cholesterol and triglycerides, is a side effect of some protease inhibitors (PIs).
Lactic Acidosis: Too much lactate in the blood and a low blood Ph (acidic) are side effects of mitochondrial toxicity, caused by of all nucleoside reverse transcriptase inhibitors (NRTIs).
Lipodystrophy: Fat redistribution, a disturbance in the way the body produces, uses and stores fat, is a side effect of the concurrent use of nucleoside reverse transcriptase inhibitors (NRTIs) and protease inhibitors (PIs).
Osteonecrosis, osteoporosis, osteopenia: Damage to the bones is a side effect of HIV and some of the damage is related to protease inhibitors (PIs).
Skin rash: NNRTIs cause most skin rashes, with Viramune causing the most severe rashes. NRTIs can also cause skin rashes.
Instant Feedback:
Hyperglycemia or diabetes is a side effect of protease inhibitors.
Instant Feedback:
Protease inhibitors (PIs) cause most skin rashes.