NSAIDs
 How NSAIDs work:
How NSAIDs work:
NSAIDs inhibit the normal
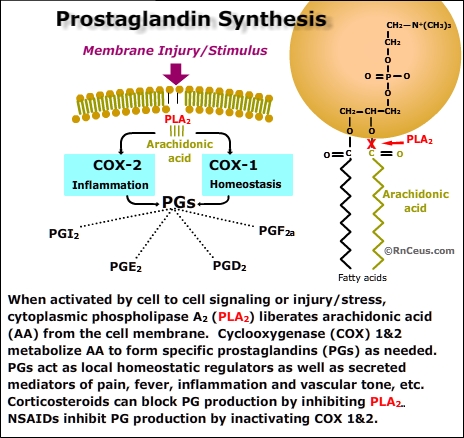
enzymatic action of the cyclooxygenase (COX) molecule. COX catalyzes the conversion of membrane fatty acids (principally arachidonic acid) to prostanoids and thromboxane. Arachadonic acid is a ubiquitous membrane fatty acid released in response to injury or cell-to-cell signaling (see panel).
Prostanoids have a short half life. They are used within the cells that produce them or are exported from the cell affecting cells close to the site of production.
- Prostanoids are involved in diverse inflammatory and homeostatic processes including:
- PG I2 - decreases platelet aggregation, increases renal blood flow, increases vasodilation, inhibits gastric acid production
- PGE2 - increases renal blood flow, mucosal protection, natriuresis, inhibits gastric acid production, pro-inflammatory, propyretic
- PGD2 - increases renal blood flow, inhibits gastric acid
production
- PGF2a - decreases progestrone, increases uterine contraction, increases vasoconstriction and bronchconstriction
- Thromboxane is responsible for platelet aggregation and vasoconstriction. Thromboxane is produced within platelets when they are exposed to collagen, thrombin, etc.•
There are two important forms of cyclooxygenase,
COX-1 and COX-2. Cells use the COX molecules to synthesize specific prostanoids in response to specific stimuli. It is simplistic to infer that COX-1 or COX-2 perform an either/or function but for our purposes:
- COX-1 is normally present
in most tissues and produces prostanoids that contribute to tissue homeostasis,
cytoprotection and cell-to-cell communication. Importantly, platelets contain only COX-1 which is irreversibly inactivate by aspirin. Platelets lack the mechanism to produce additional COX-1, therefore aspirin permanently stops the synthesis of thromboxane and thus inhibits aggregation of the exposed platelets.
- COX-2 is induced in most tissues during inflammation and injury. When nociceptors are inflammed or injured
Non-selective forms of NSAIDs
block the action of both the COX-1 and COX-2 enzymes throughout the body. Unfortunately,
inhibiting the COX-1 enzyme can produce potential life threatening complications,
particularly in the gastrointestinal tract. In the GI tract, COX-1 produces
PGI2 and PGE2 that reduce acid secretion, increase blood flow and stimulate the secretion of mucus in the esophagus, stomach, and intestine. When this protection is compromised, the
patient is at increased risk for potentially life threatening
gastrointestinal erosion, perforation and hemorrhage.
To reduce this risk, "selective" NSAIDs or "COX-2 inhibitors",
were developed and marketed. They were targeted for long term NSAID use and/or for patients
who are at risk of NSAID induced gastrointestinal or bleeding problems. NSAIDs that selectively
inhibit the COX-2 enzyme include celecoxib (Celebrex), rofecoxib (Vioxx), and
valdecoxib (Bextra).
Unfortunately, while these drugs had a greater affinity for COX-2, they were subsequently found to significantly increase the risk of potentially life threatening cardiovascular (CV) events. Inhibiting COX-2 reduced production of PGI2 by vascular endothelial cells. PGI2 has been found to be a potent vasodilator and an inhibitor of platelet aggregation. The side effects associated with COX-2 inhibitors, including hypertension and thrombotic cardiovascular events, prompted the FDA to offer the following advisory:
- Bextra (valdecoxib tablets): FDA has concluded that the overall risk versus benefit profile is unfavorable at this time and has requested the manufacturer of Bextra, Pfizer, Inc., to voluntarily withdraw Bextra from the market. This request is based on:
- the lack of adequate data on the cardiovascular safety of long-term use of Bextra, along with the increased risk of adverse CV events in short-term coronary artery bypass surgery (CABG) trials that FDA believes may be relevant to chronic use,
- reports of serious and potentially life-threatening skin reactions, including deaths, in patients using Bextra. The risk of these serious skin reactions in individual patients is unpredictable, occurring in patients with and without a prior history of sulfa allergy, and after both short- and long-term use, and
- the lack of any demonstrated advantages for Bextra compared with other NSAIDs. Pfizer has agreed to suspend sales and marketing of Bextra in the U.S. pending further discussions with the agency.
- Celebrex (celecoxib tablets): We have concluded that the benefits of Celebrex outweigh the potential risks in properly selected and informed patients. FDA has decided to allow Celebrex to remain and has asked Pfizer to take the actions listed below:
- Revise the Celebrex label to include a boxed warning containing the class NSAID warnings and contraindication about CV and GI risk, plus specific information on the controlled clinical trial data that demonstrate an increased risk of adverse CV events for celecoxib.
- Encourage practitioners to use the lowest effective dose for the shortest duration consistent with individual patient treatment goals.
- Include a Medication Guide as part of the labeling. It will be required to be given at the time the drug is dispensed to inform patients of the potential for CV and GI risk associated with NSAIDS, in general, and Celebrex specifically. The Medication Guide will inform patients of the need to discuss with their doctor the risks and benefits of using NSAIDs and the importance of using the lowest effective dose for the shortest duration possible.
- Commit to conduct a long-term study of the safety of Celebrex compared to naproxen and other appropriate drugs.
- Vioxx (rofecoxib tablets and suspension): Vioxx was voluntarily removed from the market by Merck in September 2004. FDA will carefully review any proposal from Merck for resumption of marketing of Vioxx, and would likely discuss the review with the new FDA Drug Safety Oversight Board and an Advisory Committee before making a final decision.
Instant Feedback:
Selective
NSAIDS block the action of the COX-2 enzyme.
Examples of non-selective
NSAIDs:
Aspirin
Diclofenac (Voltaren, Athrotec)
Diflunisal (Dolobid)
Etodolac (Lodine)
Fenoprofen (Nalfon)
Flurbiprofen (Ansaid)
Ibuprofen (Motrin, Advil)
Indomethacin (Indocin)
Ketoprofen (Orudis, Orudis KT)
Ketorolac (Toradol)
Mechofenamate (Meclomen)
Mefanamic acid (Ponstel)
Meloxicam (Mobic)
Nabumetone (Relafen)
Naproxyn (Naprosyn, Aleve)
Oxaproxin (Daypro)
Piroxicam (Feldene)
Sulindac (Clinoril)
Tolmetin (Tolectin)
Indications: (Oral route)
NSAID analgesics are
used for a wide variety of painful nociceptive and neuropathic conditions
of mild to moderate intensity. NSAIDS are frequently used to help relieve muscle and joint pain. If a patient is experiencing mild pain, he or
she may be given an NSAID analgesic alone. For moderate to severe pain, an NSAID analgesic
may be combined with an opioid analgesic as part of a comprehensive analgesic
plan.
Combining an opioid analgesic and a nonopioid analgesic
may provide more effective pain relief than either one alone; such drug combination
may inhibit both the transduction and the transmission of pain signals to the brain. Whenever pain is severe
enough to require an opioid analgesic, adding a nonopioid analgesic should be
considered, is a basic principle of analgesic therapy.
NSAIDs are often combined with an opioid analgesic due to
their "opioid dose-sparing" effect. Opioid dose sparing refers to
the fact that when a nonopioid is combined with an opioid, the opioid dose can
be lowered without compromising pain relief. A lower opioid dose decreases opioid
related side effects.
Instant
Feedback:
NSAIDs
are frequently used alone to treat severe pain.
Examples of oral NSAID/Opioid Combos:
- Combunox
- Schedule II controlled substance (potential for abuse)
- indicated for the short term (no more than 7 days) management of acute, moderate to severe pain
- tablet for oral administration, combines the opioid analgesic agent, oxycodone HC (5 mg), with the NSAID agent, ibuprofen (400 mg).
- WARNINGS:
CV & GI
- Serious CV and GI side effects have been associated with the use of both selective and nonselective COX 1&2 inhibitors.
- use with caution in elderly or debilitated patients, and those with severe impairment of hepatic, pulmonary or renal function, hypothyroidism, Addison's disease, acute alcoholism, convulsive disorders, CNS depression or coma, delirium tremens, kyphoscoliosis associated with respiratory depression, toxic psychosis, prostatic hypertrophy or urethral stricture.
- Percodan
- Schedule II controlled substance (potential for abuse)
- indicated for the management of moderate to moderately severe pain
- tablet for oral administration, combines the opioid analgesic agent, oxycodone HC (4.8355 mg), with the aspirin (325 mg).
- WARNINGS:
general precautions associated opioids and CV & GI precautions associated with NSAID aspirin
- not for use by children or adolescents with viral infection (Reyes syndrome) contraindication
- Ibudone
- Schedule II controlled substance (potential for abuse)
- indicated for the short-term (generally less than 10 days) management of acute pain.
- tablet for oral administration. Hydrocodone bitartrate (5 mg) and ibuprofen (200 mg)
- serious CV and GI side effects have been associated with the use of both selective and nonselective COX 1&2 inhibitors.
- use with caution in elderly or debilitated patients, and those with severe impairment of hepatic, pulmonary or renal function, hypothyroidism, Addison's disease, acute alcoholism, convulsive disorders, CNS depression or coma, delirium tremens, kyphoscoliosis associated with respiratory depression, toxic psychosis, prostatic hypertrophy or urethral stricture
Preventing and treating
NSAID side effects:
The most common side effect
of opioid analgesics is constipation, as compared to NSAIDs, which can cause
gastric ulcers, bleeding and perforation, increase bleeding time, and result
in renal insufficiency. The nonopioid acetaminophen can cause serious hepatotoxicity.
Gastrointestinal side effects associated with NSAID use can be both local and
systemic.
- Local effects occur
due to local irritation from an NSAID taken by mouth. Symptoms may include
heartburn and upper abdominal pain. These symptoms often occur early in therapy.
Local symptoms may be resolved by lowering the dose, changing to another NSAID,
taking an enteric form of an NSAID and by taking each NSAID dose with food
or a large glass of water.
- Systemic effects can
be extremely serious. Regardless of the route of administration, NSAIDs interfere with prostaglandin
synthesis throughout the entire body.
- Maintenance of the protective barrier
in the gastrointestinal tract depends on normal prostaglandin synthesis. When
this barrier is disrupted the patient is at risk of adverse events such as perforation and
hemorrhage of the esophagus, stomach, and the small or large intestine. Enteric-coated NSAIDs, food, or antacids
do not protect the patient from the risk of systemic side effects.
- Both selective and non-selective NSAIDS have been associated with Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis
- These
potentially life-threatening side effects can occur at any time during therapy,
most often without any warning signs.
Some patients are at higher than normal risk of experiencing life-threatening
gastrointestinal complications from taking NSAIDs. Those individuals at highest
risk include those who have the following characteristics:
- decreased kidney or liver function, or an uncontrolled liver problem (such as hepatitis)
- ulcer, gastritis (inflammation of the stomach lining) or stomach bleeding now or in the past
- low platelet count
- Crohn's disease or ulcerative colitis
- asthma or chronic lung disease
- reflux disease (also known as GERD), indigestion or hiatal hernia
- high blood pressure, congestive heart failure or a past stroke or heart attack
- allergy to aspirin, other NSAIDs, or sulfa drugs, or have nasal polyps (linked to a greater chance of NSAID allergy)
- concomitant use of anticoagulants or corticosteroids
- pregnant, may become pregnant, or are breast-feeding
- drink more than seven alcoholic drinks per week or more than two in a day
- age over 65
To minimize the risk of
gastrointestinal complications, NSAIDs should be used at the lowest effective
dose for the shortest time they are needed. In addition, alcohol use while taking
NSAIDs should be restricted. Patients taking NSAIDs should be advised to eat
regularly and to avoid fasting because fasting can increase toxicity. Nonselective
NSAIDs are inexpensive and usually appropriate for short term or intermittent
use. For patients who use NSAIDs on a regular basis, the more expensive but
safer selective NSAIDs may be more appropriate.
Patients who are at high
risk of adverse gastrointestinal complications may be given drugs such
as misoprostal (Cytotec), that help protect the gastrointestinal tract from
the effects of NSAIDs. NSAIDs can also cause liver damage, which is usually
detected by an increase in liver enzymes. Both nonselective and selective NSAIDs
can cause renal insufficiency. Therefore, patients who take NSAIDs should be
monitored carefully for renal impairment. Signs of renal insufficiency may include
the sudden development of oliguria with sodium and water retention. NSAIDs can
also cause some central nervous system (CNS) side effects, such as a decreased
attention span or loss of short-term memory. Because nonselective NSAIDs interfere
with normal bleeding time, they should be discontinued several days to one week
before any surgical procedure. Cox-2 NSAIDs do not decrease platelet aggregation.
As a result, Celebrex may be used perioperatively and in other clinical situations
in which bleeding is a concern.
Instant
Feedback:
Fasting
increases the risk of NSAID toxicity.
General guidelines
for using NSAIDs:
Nurses can help patients achieve the benefits of NSAIDs while reducing adverse
effects by ensuring that patients follow these guidelines:
-
Don’t
take an NSAID with alcohol.
-
Don’t
take more than one type of NSAID, with the exception of a small daily dose
of aspirin for heart attack prevention.
-
Take
NSAIDs with a full glass of water or milk, with meals, or with a prescribed
antacid. Remain upright 30 minutes after administration to reduce gastric
irritation or ulcer formation.
-
Unless
contraindicated, drink 2-3 L of water a day.
-
Report
any changes in stool consistency or symptoms of gastrointestinal irritation,
any bleeding episodes, ringing in the ears, skin rashes, sudden weight gain
and decreased urine output, fever or increased joint pain.
-
Use
caution when operating machinery or when driving a car, as NSAIDs may cause
dizziness or drowsiness.
-
If
the individual is a diabetic, he or she should be aware of interactions
between specific NSAIDs and hypoglycemic agents.
-
Notify
all healthcare providers of all medications taken.
-
Keep
all medications, both prescription and over-the-counter, in a safe place
out of the reach of children.
Instant
Feedback:
Patients
should be encouraged to combine NSAIDs for optimum pain relief.
©RnCeus.com
 How NSAIDs work:
How NSAIDs work: